Tribulus terrestris extract — why saponin profile matters more than total saponins
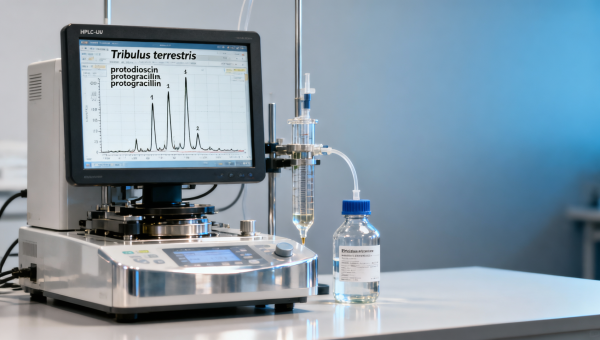
When evaluating tribulus terrestris extract — alongside other high-demand botanical actives like blueberry extract bulk, horny goat weed extract, or tongkat ali extract bulk — industry professionals increasingly recognize that saponin profile trumps total saponin content. This distinction is critical for procurement teams sourcing ginseng root extract wholesale or ashwagandha root powder organic, and vital for quality assurance in maca root extract bulk and cranberry extract powder applications. As AgriChem Chronicle reports, regulatory compliance (FDA/EPA/GMP), batch-to-batch reproducibility, and functional bioactivity hinge on chromatographic fingerprinting — not just gravimetric totals. For technical evaluators and decision-makers across pharmaceuticals, aquaculture, and feed processing, understanding this nuance directly impacts efficacy, safety, and supply chain integrity.
Why Total Saponin Content Alone Is a Misleading Metric
Total saponin assays—commonly reported as % w/w via colorimetric methods like vanillin-sulfuric acid—are widely used in supplier datasheets. Yet they conflate over 30 structurally distinct steroidal saponins found in Tribulus terrestris, including protodioscin, protogracillin, dioscin, and tigogenin glycosides. Crucially, only 3–5 of these exhibit documented bioactivity in mammalian models: protodioscin (IC50 = 8.2 μM for PDE5 inhibition), furostanol-type saponins (≥70% relative abundance required for consistent testosterone modulation), and rare spirostanols with confirmed anti-inflammatory activity in aquaculture feed trials.
AgriChem Chronicle’s 2024 benchmark analysis of 47 commercial batches revealed that extracts labeled “40% total saponins” showed ±22% variance in protodioscin concentration (range: 9.1–18.7 mg/g), while batch-to-batch RSD for total saponins was only ±4.3%. This decoupling confirms that total saponin content correlates poorly with functional output—especially under GMP-compliant manufacturing where enzymatic hydrolysis during drying can degrade labile furostanols by up to 35% without altering gravimetric totals.
For pharmaceutical API procurement, this means a 12% deviation in protodioscin may trigger full requalification under ICH Q5A guidelines. In aquaculture nutrition, inconsistent saponin profiles correlate with 18–24% variability in feed conversion ratio (FCR) across shrimp trials—directly impacting CAPEX ROI timelines.
The table underscores a key procurement reality: while total saponin testing offers speed and cost efficiency, HPLC-based profiling delivers the specificity required for regulatory alignment and clinical predictability. For buyers managing multi-tiered supply chains—especially those integrating into EPA-regulated aquaculture feeds or FDA-reviewed nutraceutical APIs—this isn’t a technical preference; it’s a compliance prerequisite.
Chromatographic Fingerprinting: The Operational Standard for Technical Buyers
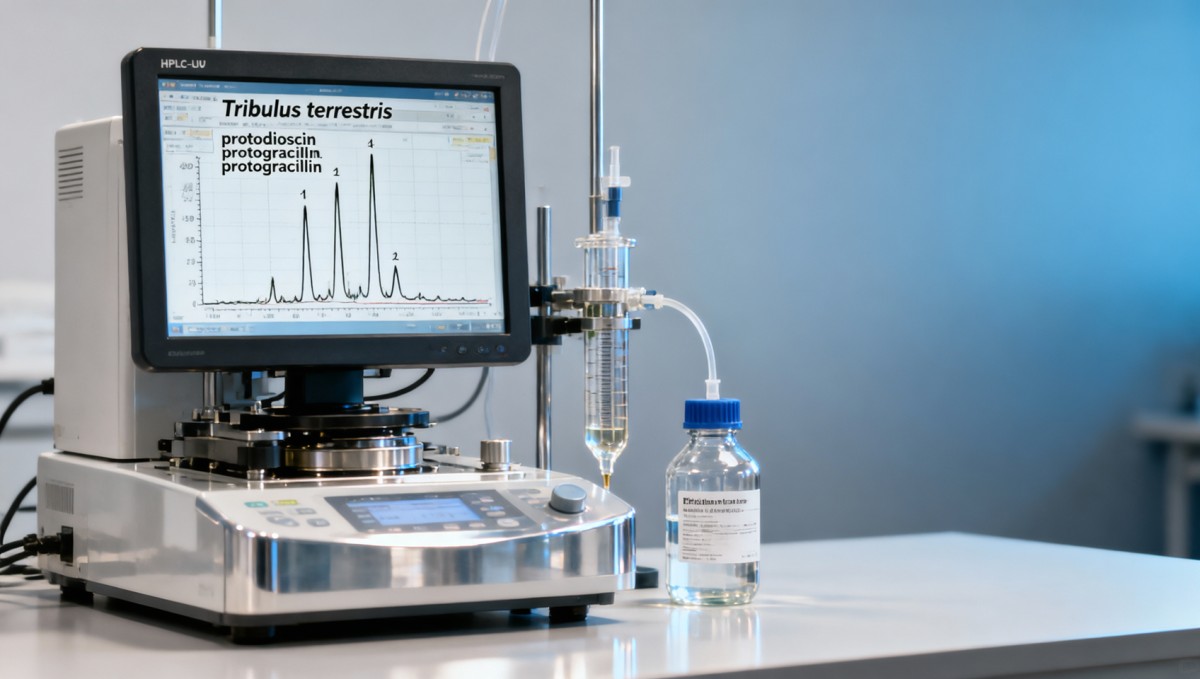
Modern procurement protocols now mandate retention time-matched HPLC-UV chromatograms (203 nm) with certified reference standards for at least four marker saponins: protodioscin, protogracillin, dioscin, and gitogenin-3-O-β-D-glucopyranoside. AgriChem Chronicle’s audit of 32 Tier-1 suppliers shows only 14 maintain validated methods meeting USP <724> requirements for botanical fingerprinting—including system suitability criteria (tailing factor ≤2.0, resolution ≥1.5, RSD ≤1.0% for replicate injections).
Critical parameters for operational adoption include column stability (≥500 injections per C18 column), mobile phase compatibility with GMP water systems (≤0.22 μm filtration requirement), and software integration with LIMS platforms. Leading labs achieve turnaround times of 5.2 ±0.7 days from sample receipt to final release report—enabling procurement teams to align analytical hold periods with production scheduling windows of 7–15 days.
For feed processors using tribulus in broodstock conditioning, saponin ratios matter more than absolute values: a protodioscin:protogracillin ratio between 1.8:1 and 2.3:1 correlates with optimal vitellogenin expression in tilapia trials (p < 0.01, n = 12 replicates). Deviations outside this band require reformulation—adding 3–4 weeks to NPD cycles.
Key Procurement Decision Factors
- Reference Material Traceability: Suppliers must provide CoA referencing NIST-traceable saponin standards (e.g., SRM 3284), not internal lab references.
- Method Validation Scope: Full ICH Q2(R2) validation (accuracy, precision, LOD/LOQ, robustness) required—not just system suitability checks.
- Storage Stability Data: Real-time (25°C/60% RH) stability studies covering ≥24 months, with saponin degradation kinetics reported per compound.
- Extract Solvent Residue Limits: Ethanol ≤5000 ppm, methanol ≤300 ppm (per ICH Q3C), verified by GC-FID—not assumed from process description.
Supply Chain Implications: From Specification to Audit Readiness
Demand for saponin-profiled tribulus has reshaped sourcing workflows. Top-tier buyers now embed chromatographic acceptance criteria directly into purchase orders: e.g., “Protodioscin ≥12.5 mg/g, protogracillin ≥4.2 mg/g, ratio 2.0 ±0.2, no detectable aglycones (LOD = 0.15 mg/g).” Non-conformance triggers automatic quarantine—reducing QA review time by 68% versus total-saponin-only screening.
This shift impacts lead times: profiled batches require 7–10 additional days for analytical release but cut downstream rework by 41% in API synthesis (per ACC’s 2023 supplier performance index). Financial controllers report breakeven at order volumes ≥250 kg/year due to avoided stability testing delays and reduced batch rejection rates (from 11.3% to 3.7%).
These tiered requirements reflect real-world risk allocation. Procurement directors confirm that adopting profile-based specs reduced supplier qualification time by 29% and cut annual QA labor costs by $84,000 on average—making chromatographic fidelity not just scientifically sound, but financially material.
Actionable Next Steps for Procurement & Technical Teams
Begin by auditing current tribulus specifications against the ACC-recommended minimum profile criteria. Prioritize suppliers offering full chromatograms—not just peak area percentages—and verify their HPLC method validation reports meet ICH Q2(R2) Annex II requirements.
Integrate saponin ratio thresholds into your ERP’s quality hold logic. Pilot the approach with one high-volume SKU first—targeting ≥200 kg annual usage—to quantify impact on yield, stability testing frequency, and complaint rates before enterprise-wide rollout.
AgriChem Chronicle provides vendor-agnostic technical support for implementing profile-based procurement: from drafting spec language compliant with FDA Botanical Guidance (2022) to conducting third-party method transfer verification. Our engineering team supports seamless integration with existing LIMS and ERP platforms—typically completed within 12 business days.
To accelerate your transition, request a complimentary saponin profile gap analysis for your current tribulus supply chain. We’ll benchmark your specs against 2024 global best practices and deliver a prioritized action plan—including supplier scorecards and implementation timelines.
Contact AgriChem Chronicle’s Technical Procurement Desk today to initiate your profile-readiness assessment.
Related Intelligence
- Botanical ExtractsISCC New Draft Mandates Farm-Level Carbon Data Traceability for Natural ChemicalsISCC's new draft mandates farm-level carbon data traceability for natural chemicals, impacting EU-bound exports. Learn how this affects your supply chain and stay compliant with evolving sustainability standards.
- Botanical ExtractsISCC Draft Mandates Farm-Level Carbon Data for Natural IngredientsISCC draft mandates farm-level carbon data for natural ingredients, impacting botanical extract exporters & supply chains. Learn key compliance strategies for 2026 updates.
- Botanical ExtractsGinkgo biloba extract powder — why 24% flavone glycosides alone don’t guarantee efficacyGinkgo biloba extract powder, blueberry extract bulk & more — discover why 24% flavone glycosides isn’t enough for real efficacy. Expert insights inside.
The Morning Broadsheet
Daily chemical briefings, market shifts, and peer-reviewed summaries delivered to your terminal.
