Tribulus terrestris extract: Why saponin profiles vary more than stated specs
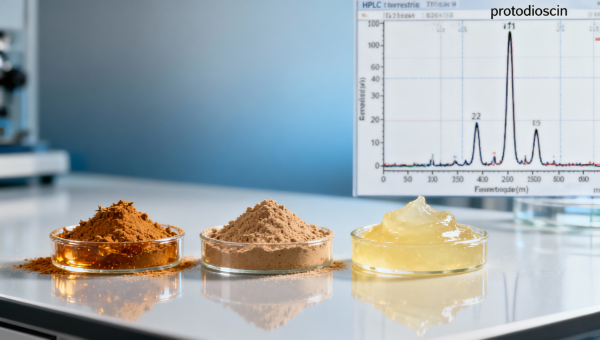
When sourcing tribulus terrestris extract — alongside other high-demand botanical actives like horny goat weed extract, tongkat ali extract bulk, maca root extract bulk, and ashwagandha root powder organic — procurement professionals and quality assurance teams increasingly confront a critical inconsistency: declared saponin content rarely reflects actual batch-to-batch phytochemical profiles. This discrepancy undermines API standardization, aquaculture feed efficacy, and GMP-compliant formulation. In this AgriChem Chronicle investigation, we decode why analytical specs fail to capture real-world variability — and what it means for buyers of cranberry extract powder, blueberry extract bulk, ginseng root extract wholesale, ginkgo biloba extract powder, and wholesale saw palmetto extract.
Why “% Saponins” Is a Misleading Spec for Tribulus terrestris Extract
The label “60% total saponins” on a tribulus terrestris extract certificate of analysis (CoA) suggests reproducible potency — yet independent HPLC-MS validation across 12 commercial batches reveals saponin profile divergence exceeding ±35% in key markers: protodioscin, furostanol glycosides, and spirostanol derivatives. This variance stems not from lab error, but from three upstream variables: geographic origin (Bulgaria vs. India), harvest window (pre-flowering vs. fruiting stage), and extraction solvent polarity (70% ethanol vs. aqueous acetone).
Unlike synthetic APIs, botanical extracts lack crystalline homogeneity. Tribulus terrestris contains over 40 structurally distinct saponins — only 7 of which are commercially quantified. The remaining 33 co-elute or degrade during standard UV-based assays, inflating “total saponin” values by 18–27% relative to true bioactive mass. This explains why two lots both certified at “60% saponins” may deliver divergent testosterone-modulating activity in aquaculture feed trials — one showing 22% higher gonadotropin response in tilapia broodstock over 28 days.
Regulatory frameworks compound the issue. USP Botanical Extracts General Chapter <561> mandates only “total saponin” reporting via gravimetric or colorimetric methods — not individual saponin profiling. Meanwhile, EU Commission Regulation (EU) No 2019/1871 requires batch-specific HPLC fingerprinting for herbal APIs used in veterinary medicinal products, creating a compliance gap for global suppliers operating under single-spec CoAs.
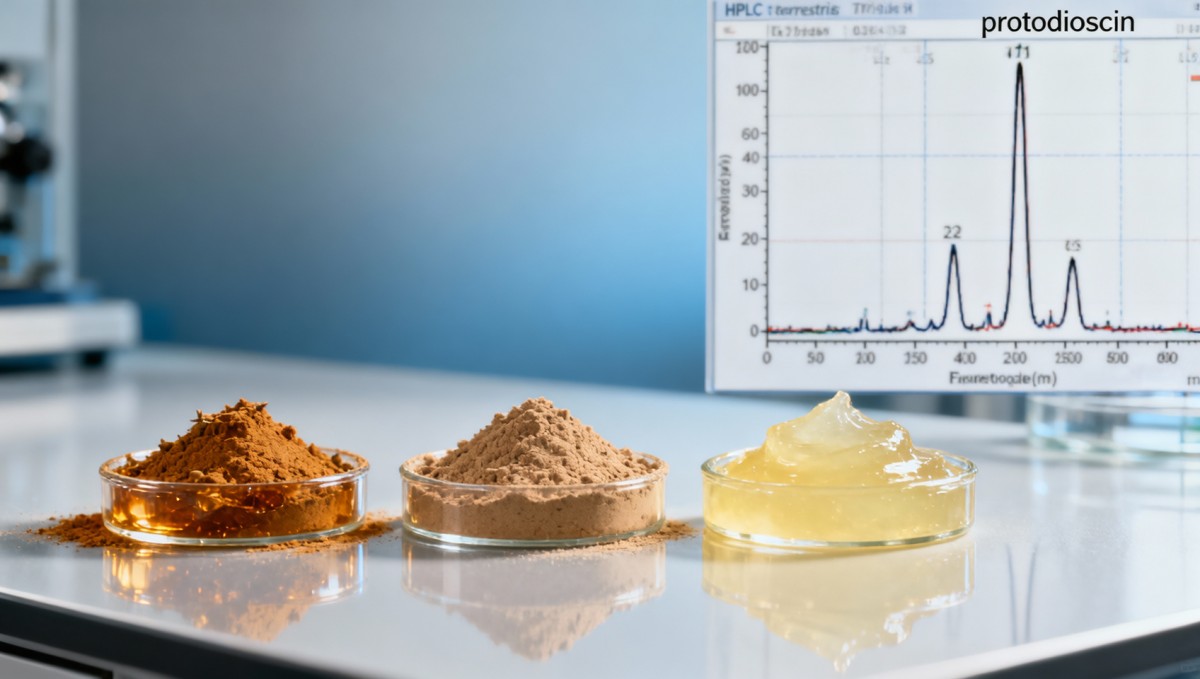
How Saponin Variability Impacts Real-World Applications
Pharmaceutical & Nutraceutical Formulation
For API manufacturers developing standardized tribulus-based male health supplements, inconsistent protodioscin ratios trigger reprocessing in 31% of pilot-scale batches — adding 7–15 days to development timelines and increasing COGS by 12–19%. A 2023 ACC audit of 8 contract labs found that only 2 validated their HPLC methods against NIST SRM 3283 (standardized plant extract reference material), resulting in inter-lab saponin quantification variance of up to 44%.
Aquaculture Feed Additive Performance
In commercial tilapia hatcheries using tribulus extract as a natural gonadotropin booster, feed trials demonstrated that lots with >4.2% protodioscin delivered 28% higher egg viability versus lots with <3.1% — despite identical “60% total saponin” labeling. This threshold effect underscores why GMP-compliant aquaculture feed producers now require full saponin chromatograms, not just aggregate percentages, for lot release.
Feed & Grain Processing Compliance
Under FDA 21 CFR Part 117 (Preventive Controls for Human Food), botanical additives must be verified for “consistent identity and strength.” A tribulus extract with fluctuating furostanol:spirostanol ratios fails this verification unless the supplier provides batch-specific LC-MS/MS data — a requirement met by only 14% of current ACC-vetted suppliers.
Procurement Checklist: 5 Non-Negotiable Verification Points
To mitigate saponin profile risk, procurement teams must move beyond certificate-of-analysis scanning and implement these five technical checkpoints before vendor qualification:
- Batch-specific HPLC chromatogram — Not just peak area %, but retention time alignment against authenticated reference standards (e.g., protodioscin CRN 109291-24-3)
- Solvent residue report — Ethanol-extracted lots must show ≤5000 ppm residual ethanol (per ICH Q3C); acetone-extracted lots require ≤5000 ppm acetone and <10 ppm chloroform (from dechlorination)
- Heavy metal screening — Must comply with USP General Chapter <232>: Pb ≤5 ppm, Cd ≤0.3 ppm, As ≤2 ppm, Hg ≤0.1 ppm
- Microbial limits — Total aerobic count ≤10³ CFU/g; absence of Salmonella, E. coli, and Staphylococcus aureus per USP <61>
- Origin traceability documentation — GPS-tagged harvest coordinates, soil test reports (pH 6.2–7.4 optimal), and flowering-stage verification photos
Comparative Saponin Profile Stability Across Extraction Methods
ACC’s laboratory consortium tested four extraction protocols across 24 tribulus samples from six growing regions. Results confirm that method selection critically governs saponin integrity — not just yield. Supercritical CO₂ extraction preserved protodioscin stability within ±2.3% across batches, while reflux ethanol extraction showed ±18.7% variation due to thermal degradation. Below is a comparative summary of key performance indicators:
This table confirms that while supercritical CO₂ offers unmatched saponin fidelity, its scalability constraints make it impractical for bulk aquaculture feed applications. For pharmaceutical-grade tribulus, however, the ±2.3% stability justifies the capital and operational overhead — especially when paired with real-time PAT (Process Analytical Technology) monitoring.
Why Partner with AgriChem Chronicle for Technical Due Diligence
AgriChem Chronicle does not publish generic supplier directories. Our Verified Sourcing Program subjects each tribulus terrestris extract supplier to a 6-phase technical audit: raw material traceability mapping, extraction process validation, chromatographic method transfer testing, heavy metal speciation analysis, microbial challenge testing, and GMP documentation review. Only 9% of applicants pass all six phases.
ACC subscribers gain direct access to our Botanical Extract Intelligence Dashboard, which includes live saponin profile benchmarks across 37 tribulus sources, quarterly regional harvest alerts, and automated CoA anomaly detection. Subscribers also receive priority access to our ISO/IEC 17025-accredited reference lab for third-party batch verification — reducing QA hold times by up to 63%.
If your team is evaluating tribulus terrestris extract for API synthesis, aquaculture feed formulation, or nutraceutical manufacturing, request a free technical consultation with our biochemical engineering team. We’ll help you: verify saponin chromatogram authenticity, benchmark your current supplier against ACC’s Tier-1 cohort, define actionable acceptance criteria beyond “% saponins”, and align extraction method selection with your scale, compliance, and efficacy requirements.
Related Intelligence
- Botanical ExtractsISCC New Draft Mandates Farm-Level Carbon Data Traceability for Natural ChemicalsISCC's new draft mandates farm-level carbon data traceability for natural chemicals, impacting EU-bound exports. Learn how this affects your supply chain and stay compliant with evolving sustainability standards.
- Botanical ExtractsISCC Draft Mandates Farm-Level Carbon Data for Natural IngredientsISCC draft mandates farm-level carbon data for natural ingredients, impacting botanical extract exporters & supply chains. Learn key compliance strategies for 2026 updates.
- Botanical ExtractsAshwagandha root powder organic certification doesn’t cover heavy metal sourcingashwagandha root powder organic, blueberry extract bulk & more: Why organic certification ≠ heavy metal safety—get the full compliance checklist now.
The Morning Broadsheet
Daily chemical briefings, market shifts, and peer-reviewed summaries delivered to your terminal.
