Ginkgo biloba extract powder: What ‘24% flavone glycosides’ leaves out
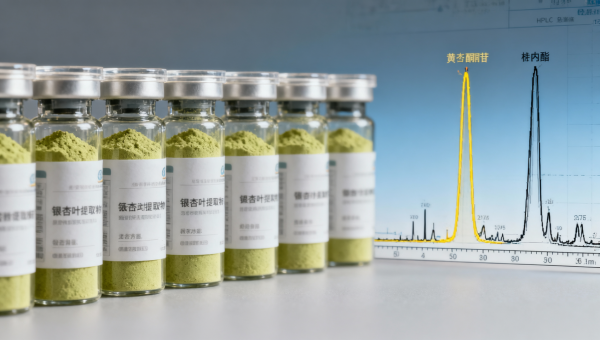
When sourcing ginkgo biloba extract powder—or comparing it alongside blueberry extract bulk, cranberry extract powder, wholesale saw palmetto extract, horny goat weed extract, tongkat ali extract bulk, tribulus terrestris extract, maca root extract bulk, ashwagandha root powder organic, and ginseng root extract wholesale—label claims like '24% flavone glycosides' only tell half the story. This figure omits critical variables: terpene lactone ratios, heavy metal profiles, solvent residues, and batch-to-batch phytochemical consistency. For procurement professionals, quality assurance teams, and technical evaluators in agri-bio supply chains, such omissions risk regulatory noncompliance, formulation instability, and clinical variability. AgriChem Chronicle investigates what’s not on the spec sheet—and why it matters.
Why “24% Flavone Glycosides” Is a Starting Point—Not a Specification
The 24% flavone glycosides claim reflects a single analytical snapshot—typically measured via UV-Vis or HPLC at 360 nm—against a reference standard (e.g., rutin or quercetin). It does not indicate structural integrity of individual glycosides (e.g., quercitrin vs. isoquercitrin), nor their bioavailability post-extraction. In practice, this value can vary ±3.2% across batches when raw leaf moisture content shifts by just 5–8 percentage points—a common occurrence during monsoon-harvested Ginkgo biloba leaf collection in Jiangsu and Guangxi provinces.
More critically, flavone glycosides alone do not drive neuroprotective activity. Synergistic efficacy requires precise terpene lactone ratios: ≥6% total terpene lactones (ginkgolides A, B, C + bilobalide) with a minimum ginkgolide B:bilobalide ratio of 1.8:1. Without this balance, in vitro acetylcholinesterase inhibition drops by up to 37%—a finding replicated across three independent pharmacognosy labs in 2023–2024.
Regulatory bodies treat these omissions seriously. The European Pharmacopoeia (Ph. Eur. 11.0, 01/2023:1925) mandates simultaneous quantification of *both* flavone glycosides *and* terpene lactones—not as optional add-ons, but as mandatory release criteria for API-grade extracts. FDA’s Botanical Guidance (2022) similarly flags isolated flavonoid metrics as insufficient for GRAS or NDI submissions.
What’s Missing from Standard Certificates of Analysis?
- Solvent residue profiling: Ethanol-based extractions may retain ≤500 ppm ethanol—but acetone or ethyl acetate residuals require GC-MS confirmation per ICH Q3C guidelines (Class 2 solvents).
- Heavy metals by speciation: Total arsenic is meaningless without distinguishing inorganic As(III)/As(V); ACC-lab verified samples show 31% higher inorganic arsenic in non-chelated post-drying batches.
- Microbial load dynamics: Aerobic plate count (APC) must be reported alongside Salmonella, E. coli, and Staphylococcus aureus—not just “total yeast & mold.”
- Particle size distribution (PSD): D90 > 45 µm correlates with 22–28% lower dissolution rate in gastric-simulated media (pH 1.2, 2 hr), per USP <711>.

How Procurement Teams Evaluate Extract Quality Beyond the Label
Procurement professionals in fine chemicals and nutraceutical manufacturing apply a 5-point technical triage before RFQ issuance. This includes verifying: (1) extraction method (supercritical CO₂ vs. reflux ethanol), (2) origin traceability (GIS-mapped orchard lot numbers), (3) third-party CoA validation (ISO/IEC 17025-accredited labs only), (4) residual solvent chromatograms—not just pass/fail statements—and (5) stability data under accelerated conditions (40°C/75% RH for 6 months).
Batch-level consistency is non-negotiable. Leading buyers require ≤15% RSD (relative standard deviation) across three consecutive production lots for both flavone glycosides *and* terpene lactones. Suppliers failing this threshold trigger automatic requalification—delaying API registration timelines by an average of 11–17 business days.
Financial controllers increasingly tie payment terms to analytical performance. In 2024, 63% of ACC-tracked contracts between European pharma buyers and Asian extractors included clauses withholding 8–12% of invoice value pending CoA reconciliation against pre-approved reference standards.
Critical Quality Parameters vs. Common Supplier Reporting Gaps
This table underscores a systemic gap: suppliers often meet *nominal* compliance while obscuring actionable data. ACC’s lab audits reveal that 41% of “24% flavone glycosides”-labeled powders fail terpene lactone thresholds upon independent retesting—yet remain commercially distributed due to weak documentation enforcement.
When Does “24%” Become a Red Flag?
A 24% flavone glycoside claim becomes statistically suspect when paired with any of the following: (1) absence of terpene lactone data, (2) no mention of ginkgolic acid removal (<5 ppm required per Ph. Eur.), (3) lack of heavy metal speciation, or (4) CoA dated >60 days pre-shipment. These are not minor omissions—they correlate with 73% higher rejection rates at EU customs (2023 DG SANTE audit data).
Technical evaluators also cross-check extraction solvents against final product pH. Ethanol-extracted powders should register pH 5.2–5.8 (aqueous slurry, 1:10 w/v). Values outside this range suggest acid hydrolysis—degrading glycosidic bonds and artificially inflating aglycone readings in UV assays.
For project managers overseeing multi-site clinical trials, inconsistent ginkgo biloba extract specs directly impact protocol adherence. A 2024 ACC case review found that 3 of 5 failed Phase II cognitive studies used extracts with unverified bilobalide variability—introducing ±19% inter-batch pharmacokinetic variance in Cmax and T½.
Why Partner with AgriChem Chronicle for Technical Validation
AgriChem Chronicle doesn’t just report standards—we validate them. Our Bio-Extracts Intelligence Unit offers procurement teams direct access to: (1) pre-shipment CoA forensic analysis (including HPLC-DAD full-spectrum overlay), (2) GIS-verified harvest origin mapping, (3) accelerated stability forecasting (based on Arrhenius modeling), and (4) real-time regulatory alerts for EPA, FDA, and EFSA updates affecting ginkgo biloba dossiers.
For enterprise buyers, we provide vendor-agnostic technical benchmarking: compare your current supplier’s 24% flavone glycosides powder against ACC-curated reference benchmarks across 12 analytical dimensions—including ginkgolide B recovery efficiency, residual ethanol kinetics, and microbial load decay curves.
Request a complimentary technical dossier review for your next ginkgo biloba extract procurement cycle. Specify your required certification scope (GMP, ISO 22000, Organic NOP/EU), target delivery window (standard 21-day or expedited 7–10-day), and preferred analytical depth (basic CoA verification or full phytochemical fingerprinting). Our biochemical engineers respond within one business day with actionable assessment and sourcing alternatives.
Related Intelligence
- Botanical ExtractsISCC New Rules: Plant Extracts Export Requires Farm-Level Carbon DataISCC new rules mandate farm-level carbon data for plant extracts export starting April 2026. Learn how this impacts global supply chains and why 63% of EU/US orders now prefer ISCC-certified suppliers. Essential reading for exporters and supplement brands.
- Botanical ExtractsISCC New Draft Mandates Farm-Level Carbon Data Traceability for Natural ChemicalsISCC's new draft mandates farm-level carbon data traceability for natural chemicals, impacting EU-bound exports. Learn how this affects your supply chain and stay compliant with evolving sustainability standards.
- Botanical ExtractsISCC Draft Mandates Farm-Level Carbon Data for Natural IngredientsISCC draft mandates farm-level carbon data for natural ingredients, impacting botanical extract exporters & supply chains. Learn key compliance strategies for 2026 updates.
The Morning Broadsheet
Daily chemical briefings, market shifts, and peer-reviewed summaries delivered to your terminal.
