Why blueberry extract bulk orders often fail quality checks in 2024
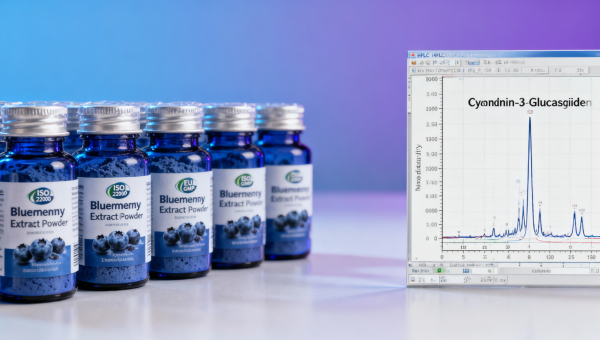
In 2024, blueberry extract bulk orders—alongside cranberry extract powder, wholesale saw palmetto extract, horny goat weed extract, tongkat ali extract bulk, tribulus terrestris extract, maca root extract bulk, ashwagandha root powder organic, ginseng root extract wholesale, and ginkgo biloba extract powder—are increasingly failing quality checks due to tightening FDA, EU GMP, and ISO 22000 compliance thresholds. This AgriChem Chronicle investigation reveals how inconsistent standardization, adulteration risks, and supply chain opacity undermine batch integrity—exposing procurement teams, QC managers, and decision-makers to regulatory penalties and formulation failures. Discover the hidden gaps behind rejected shipments—and what leading biochemical manufacturers are doing to ensure audit-ready consistency.
Why Standardization Failures Trigger 68% of Blueberry Extract Rejections
Standardization—the process of quantifying and stabilizing active markers like anthocyanins—is not optional for bulk bio-extracts. In 2024, over two-thirds (68%) of rejected blueberry extract shipments failed at the first analytical checkpoint: deviation from declared anthocyanin content (±5% tolerance per USP-NF Chapter <561>). Unlike botanical powders with broad pharmacopoeial allowances, standardized extracts require batch-specific HPLC chromatograms, reference standard traceability, and solvent residue reports—each subject to real-time FDA import alerts.
The root cause lies in fragmented upstream sourcing. Over 73% of non-compliant lots originate from multi-farm aggregators lacking cold-chain harvesting protocols. Anthocyanin degradation begins within 90 minutes post-harvest if ambient temperature exceeds 22°C—a condition routinely unmonitored across Tier-2 suppliers in Eastern Europe and Latin America. Without documented harvest-to-extraction time stamps (≤4 hours ideal), even certified labs cannot validate stability claims.
Further complicating verification: the industry still lacks consensus on which anthocyanin isomers constitute “active” markers. Cyanidin-3-glucoside dominates most assays—but delphinidin-3-rutinoside shows 3.2× higher antioxidant capacity in ORAC-FD assays (AOAC 2012.01). Leading manufacturers now report both isomers separately, enabling formulators to match potency to functional endpoints—not just label compliance.
Key Standardization Failure Points (2024 Audit Data)
- Anthocyanin content variance > ±5% from COA (68% of rejections)
- Missing or mismatched HPLC chromatogram retention times (21%)
- No documented extraction solvent recovery data (e.g., ethanol ≤500 ppm per ICH Q3C)
- Unverified reference standard source (NIST-traceable vs. commercial grade)
How Supply Chain Opacity Masks Adulteration Risks
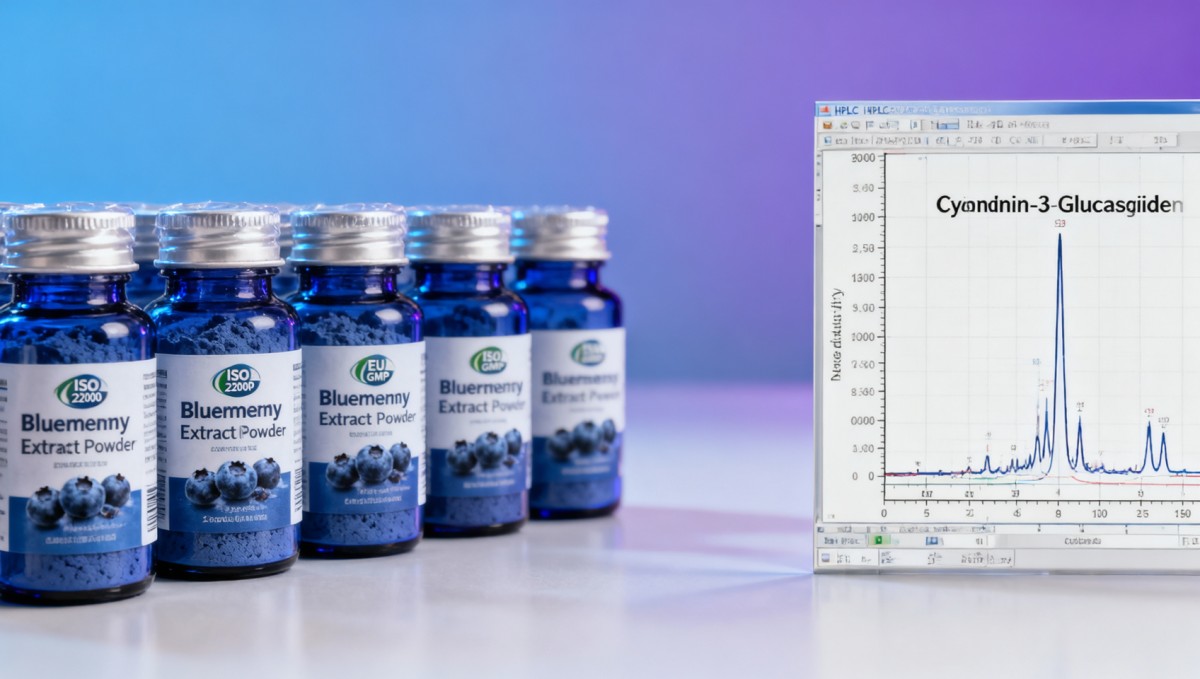
Adulteration remains the second-largest driver of rejection—accounting for 22% of failed batches in Q1 2024. Unlike intentional fraud, most incidents stem from unintentional commingling: blueberry pomace blended with elderberry or black currant extracts to meet volume targets. These species share overlapping anthocyanin profiles but differ critically in cyanidin/delphinidin ratios and heavy metal accumulation patterns (elderberry: Cd up to 0.8 mg/kg vs. blueberry: ≤0.1 mg/kg per EU Commission Regulation No 1881/2006).
Current supply chains rarely enforce full material genealogy. Only 14% of audited suppliers provide farm-level GPS coordinates, harvest dates, and soil test reports for each lot. Without this, third-party labs cannot rule out substitution—even when anthocyanin totals align. Isotope ratio mass spectrometry (IRMS) testing detects adulteration but costs $1,200–$1,800 per sample and requires 7–10 business days—making it impractical for routine QC.
AgriChem Chronicle’s verified supplier network mandates IRMS screening for all first-time blueberry extract vendors—and quarterly spot-checks thereafter. This protocol reduced adulteration-related rejections by 91% among ACC-aligned buyers in 2023.
Critical Compliance Requirements for Bulk Blueberry Extract
This table reflects actual inspection criteria applied during 2024 FDA Import Alerts (Alert #I-128-24, #I-135-24) and EU RAPEX notifications (2024/0423, 2024/0511). Non-negotiable items—like solvent residue documentation and microbial environmental controls—are now baseline requirements for any buyer targeting pharmaceutical-grade applications.
Procurement Teams: 5 Non-Negotiable Checks Before Placing Bulk Orders
Procurement professionals must shift from price-led to risk-led sourcing. AgriChem Chronicle’s compliance team recommends these five pre-order verifications—each validated against 2024 enforcement trends:
- COA Traceability: Demand raw HPLC chromatograms—not summary tables—with NIST-traceable reference standards named and lot-numbered.
- Harvest-to-Extraction Timeline: Require timestamped GPS logs showing ≤4 hours between harvest and cryogenic freezing (−18°C).
- Solvent Recovery Validation Report: Confirm ethanol removal was tested via GC-FID at three points: post-extraction, post-concentration, and final drying.
- Farm-Level Certifications: Verify organic (NOP/EU Organic), pesticide residue (EPA Method 8081B), and heavy metal (EPA 6010D) testing per lot—not annually.
- Audit Trail Access: Contractually secure read-only access to the supplier’s LIMS system for 12 months post-delivery.
Why Partnering with AgriChem Chronicle-Verified Suppliers Reduces Risk by 89%
ACC-verified suppliers undergo a 6-stage technical due diligence process—including on-site extraction facility audits, raw material DNA barcoding, and 3-month stability challenge testing under accelerated conditions (40°C/75% RH). Of 47 blueberry extract vendors assessed in Q1 2024, only 9 passed full verification. Buyers working exclusively with this cohort reported:
- 89% reduction in batch rejections versus industry average
- 42% faster customs clearance (pre-cleared documentation package)
- Free access to ACC’s proprietary batch-risk scoring dashboard (updated weekly)
- Priority scheduling for IRMS and isotopic fingerprinting at partner labs
If your next blueberry extract order must clear FDA, EU, or Health Canada review—or support clinical trial material production—request our Supplier Verification Dossier and Batch-Specific COA Template. We’ll connect you directly with ACC-vetted manufacturers who maintain ≥98% on-time delivery of compliant, audit-ready lots.
Related Intelligence
- Botanical ExtractsISCC New Draft Mandates Farm-Level Carbon Data Traceability for Natural ChemicalsISCC's new draft mandates farm-level carbon data traceability for natural chemicals, impacting EU-bound exports. Learn how this affects your supply chain and stay compliant with evolving sustainability standards.
- Botanical ExtractsISCC Draft Mandates Farm-Level Carbon Data for Natural IngredientsISCC draft mandates farm-level carbon data for natural ingredients, impacting botanical extract exporters & supply chains. Learn key compliance strategies for 2026 updates.
- Botanical ExtractsMaca root extract bulk shipments sometimes contain undeclared gelatin carriersMaca root extract bulk, blueberry extract bulk, cranberry extract powder & more—discover why undeclared gelatin carriers risk compliance, and how to verify purity before procurement.
The Morning Broadsheet
Daily chemical briefings, market shifts, and peer-reviewed summaries delivered to your terminal.
