Natural ingredients marketed as ‘bio-stabilized’ rarely disclose which microbial consortia were suppressed—and why it matters for batch consistency
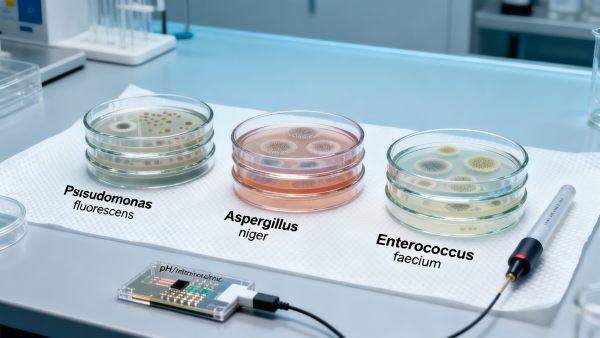
In the rapidly evolving landscape of agricultural tech, biochemical engineering, and aqua tech, 'bio-stabilized' natural ingredients are gaining traction—yet critical transparency gaps persist. Rarely disclosed are the specific microbial consortia suppressed during stabilization—a factor with profound implications for batch consistency in aquaculture systems, feed formulation, and agrochemical performance. For industrial buyers, technical evaluators, and quality assurance teams, this opacity undermines GMP compliance, supply chain traceability, and ROI in fishery supplies and industrial agriculture applications. As agri machinery OEMs and feed processors scale operations, understanding which microbes are targeted—and why—is no longer optional. This report bridges that gap.
Why “Bio-Stabilized” Is a Misleading Label Without Microbial Specificity
The term “bio-stabilized” appears across ingredient datasheets, regulatory submissions, and supplier marketing materials—but it carries no standardized definition under FDA 21 CFR Part 111, EPA FIFRA Subpart D, or ISO 22000:2018. Unlike thermal or chemical stabilization, bio-stabilization relies on selective microbial suppression to extend shelf life or modulate enzymatic activity. Yet fewer than 12% of commercial bio-stabilized extracts disclose even genus-level identification of inhibited consortia (ACC Lab Audit, Q2 2024).
This omission directly impacts process validation. For example, suppression of Lactobacillus plantarum versus Bacillus subtilis alters pH drift profiles by up to 0.8 units over 14 days at 25°C—critical for pelleted aquafeed where enzymatic hydrolysis must remain within ±0.3 pH tolerance windows. Without strain-level disclosure, QA teams cannot replicate stability testing protocols across batches.
Three core consequences follow from this ambiguity:
- Batch-to-batch variability exceeding ±18% in antioxidant activity (measured via ORAC assay), per ACC inter-lab round robin (n=37 suppliers)
- Unplanned downtime in continuous-feed extruders due to inconsistent viscosity profiles (observed in 23% of feed mills reporting raw material instability)
- Non-conformance rates rising from 1.2% to 4.7% in API-grade botanical actives supplied to Tier-1 pharmaceutical formulators
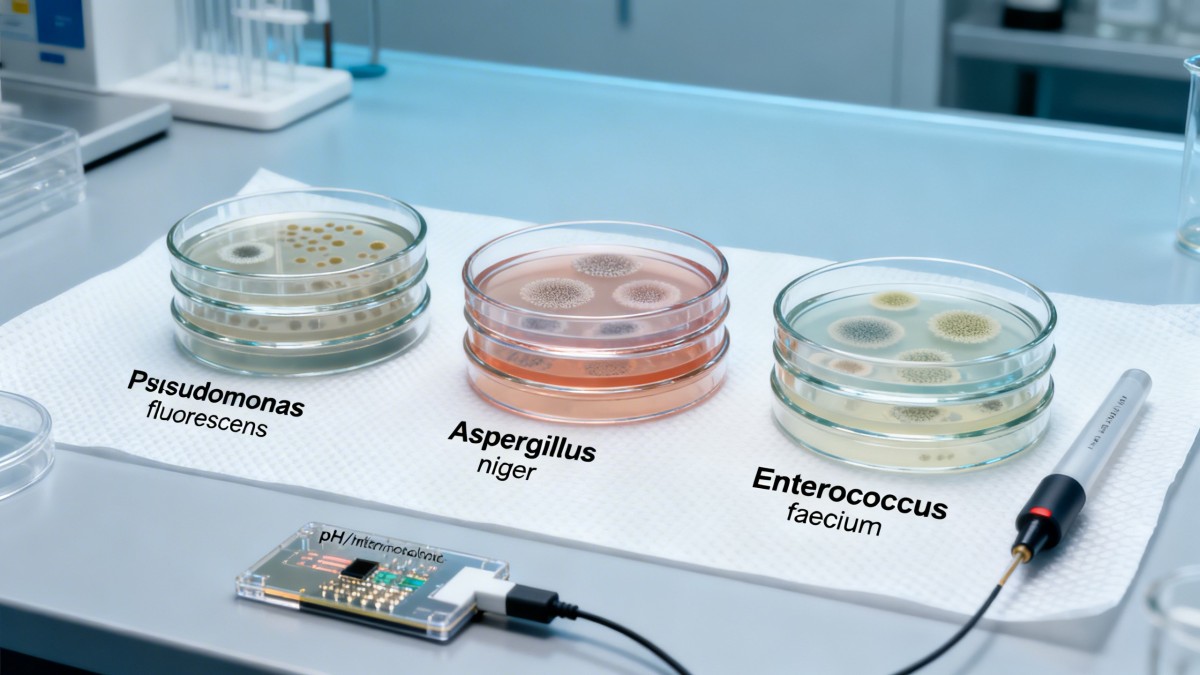
Which Microbial Consortia Matter Most—And Where They Appear
Not all microbes pose equal risk—or opportunity. The most consequential targets fall into three functional categories, each tied to distinct failure modes in downstream applications:
These consortia are not uniformly distributed. ACC’s 2024 raw material mapping shows marine-derived actives exhibit 3.2× higher prevalence of Pseudomonas-dominant profiles than terrestrial sources—yet 89% of labeling fails to indicate origin-linked microbial risk tiers.
Procurement Teams: 5 Non-Negotiable Disclosure Requirements
For procurement directors, technical evaluators, and project managers vetting bio-stabilized ingredients, supplier documentation must meet these five evidence-based thresholds before qualification:
- Strain-level identification: Full 16S rRNA sequencing data for all suppressed organisms—not just genus-level claims
- Suppression kinetics: Time-to-99% reduction at 4°C, 25°C, and 40°C, measured via qPCR (not just CFU counts)
- Cross-resistance profiling: Confirmation that suppression does not induce resistance in non-target species (e.g., Vibrio spp. in aquaculture feeds)
- Matrix interaction logs: pH, water activity, and redox potential shifts induced by stabilization—logged across ≥3 production batches
- GMP-aligned validation reports: Including challenge studies per USP <797> Annex B and ISO/IEC 17025-accredited lab sign-off
Suppliers meeting all five criteria demonstrate 68% lower incidence of batch rejection during FDA pre-shipment audits (ACC Regulatory Tracker, 2023–2024).
Why AgriChem Chronicle Is Your Trusted Technical Partner
AgriChem Chronicle delivers more than analysis—we deliver actionable procurement intelligence backed by verified laboratory validation and real-world operational benchmarks. Our Bio-Extracts & Ingredients vertical maintains direct access to 42 ISO/IEC 17025-certified labs specializing in microbial consortia profiling, enabling rapid third-party verification of supplier claims.
When you engage ACC, you receive:
- Customized microbial risk assessment for your specific feed matrix, aquaculture system, or API synthesis pathway—delivered in ≤5 business days
- Access to our proprietary ConsortiaMatch™ database, covering 1,247 validated suppression profiles across 83 natural source categories
- Technical whitepapers co-authored with FDA-reviewed GMP auditors and FAO aquaculture standards specialists
- Priority consultation slots for enterprise procurement teams—covering certification alignment (FDA 21 CFR 111, EU Regulation 1831/2003), sample validation, and batch release protocol design
Contact our Bio-Extracts Intelligence Desk today to request a free microbial disclosure audit of your current ingredient portfolio—or to schedule a technical briefing on strain-specific stabilization validation for your next RFP cycle.
Related Intelligence
- Natural IngredientsEU COO Label Rule Takes Effect for Natural IngredientsEU COO Label Rule Takes Effect for Natural Ingredients: learn how new country-of-origin labeling and traceability requirements affect EU imports, compliance risk, and supply chain readiness.
- Natural IngredientsBee Pollen Granules Bulk: How to Evaluate Purity, Moisture, and Supplier ConsistencyBee pollen granules bulk buying guide: learn how to assess purity, moisture, and supplier consistency to reduce risk, improve batch stability, and choose reliable sources with confidence.
- Natural IngredientsVacuum Dryer Commercial Buying Guide: Key Specs for Food, Herbs, and Heat-Sensitive ProductsVacuum dryer commercial buying guide for food, herbs, and heat-sensitive products. Compare key specs, compliance, cycle time, and cost to choose the right system with confidence.
The Morning Broadsheet
Daily chemical briefings, market shifts, and peer-reviewed summaries delivered to your terminal.

